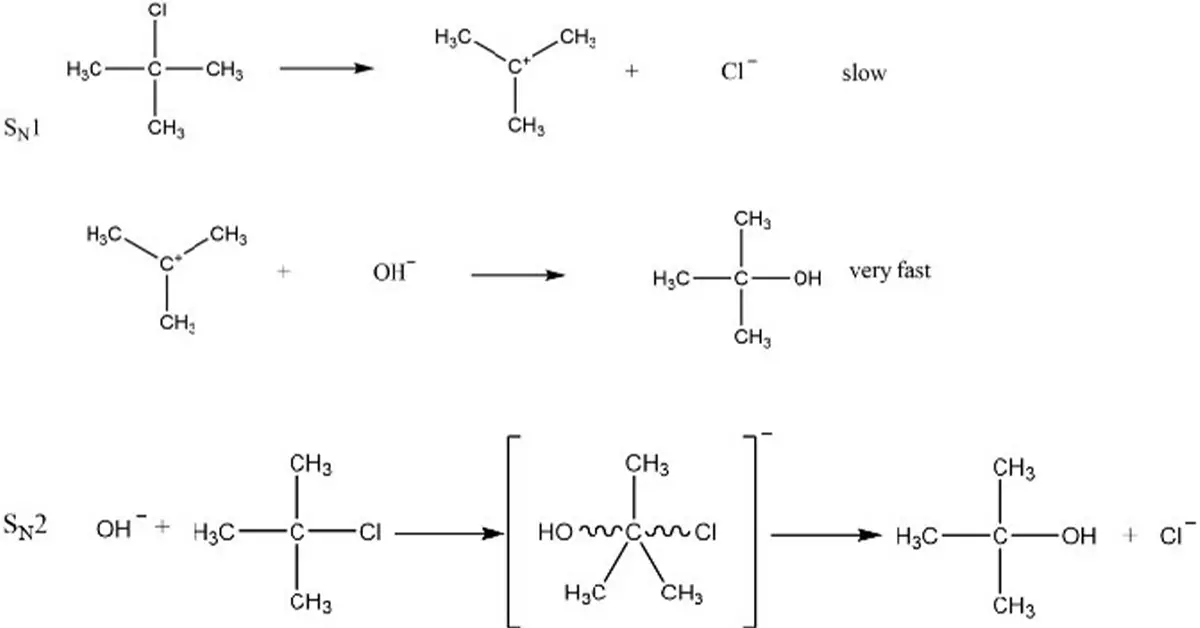
There are many similarities between sn1 and sn2, but there are also some key differences. The most important difference is the way they react with water. The hydrogen in sn1 reacts with water to make hydrogen gas and leaves behind the other elements in the salt, which is why it’s called a “strong” base. On the other hand, when you add water to an ice cube made of sn2 – that contains sodium chloride – nothing happens! This is because both hydrogen and oxygen have been removed from this substance so there isn’t anything for them to react with. It’s called a “weak” base because it doesn’t interact very well with water.
The difference between sn1 and sn2 is that the latter has an additional hydrogen atom. The reason for this is that one of the oxygen atoms in the molecule got replaced with a sulfur atom. This means that there are two more lone pairs on carbon, which will give it a higher polarity than its predecessor because it can form stronger bonds to other molecules. There are many benefits to using this type of compound over others, such as creating less toxic waste or having better solubility than some others. One downside though is how much energy it takes to produce them. It could be worth exploring alternate routes for production if possible so we can find solutions to these challenges while still benefiting from their properties.
Comparison between sn1 and sn2
| Parameters of Comparison | sn1 | sn2 |
| Shape | Shape is different | Shape different |
| Bond | Single bond | Double bond |
| Weight | Different weight | Different weight |
| Suger | Different Suger | Different Suger |
| Known | sn1 is also known as dextrose or grape sugar | sn2 is called glucose |
What is sn1?
Sn1 stands for the first step in a chemical reaction. The sn1 is also called an “initiator.” This means that it starts the process of the reaction. It’s important to know this because it can help you understand how reactions are happening. For example, if you have sugar and water, there will be no chemical changes until someone adds an initiator like heat or acid. Then, there will be a new substance formed from these two things: CO2 and H2O (which is what we call water). If somebody added vinegar instead of heat or acid then acetate would form (CH3COOCOOH) with sugar and water as well as some other substances forming during this transformation process. Sn1 stands for the first step in a reaction when one molecule has been substituted by another. To know if a reaction is Sn1, you need to look at the reactants and see which atom of each reactant has been replaced with that of the other. If only one atom was changed then it is an Sn1 reaction. This can be seen in many reactions including ones involving hydrolysis, decarboxylation, halogenations, and oxidations.

What is sn2?
The SN2 mechanism is an organic reaction involving the nucleophilic substitution of an alkyl halide with another nucleophile. It is one of the most powerful reactions in organic synthesis because it can be used to make carbon-carbon bonds, create new stereocenters and introduce functional groups into molecules. The SN2 mechanism has two steps: first, there’s a nucleophilic attack on the substrate (the alkyl halide) by a base; then elimination occurs to afford either backside or frontside departure of the leaving group (X).
The SN2 mechanism doesn’t work for tertiary substrates because this would cause ring strain due to poor overlap between adjacent atoms. However, if you turn your product into a better nucleophile (eg: by adding an electronegative atom) then the SN2 will work with tertiary substrates.
10 Differences Between sn1 and sn2
1. The first difference is that the two molecules have different shapes.
2. Another difference is that sn1 has a single bond and sn2 has double bonds.
3. A third difference between these molecules is that they have different molecular weights.
4. Lastly, one more big difference between them is their boiling points – which are higher for sn1 than for sn2.
5. One of the most important things to remember about these two molecules is that they can both exist as gases under certain conditions and also as solids at room temperature depending on what kind of molecule it’s bonded with.
6. sn1 and sn2 are different types of sugars.
7. sn1 is also known as dextrose or grape sugar, while sn2 is called glucose.
8. The molecular structure of the two sugars differs – in the case of dextrose, it has a right-handed helix, whereas in glucose it’s left-handed.
9. Sn1 can be found naturally in fruits such as grapes and apples, but not so much in other types of fruit like bananas or oranges.
10. Sn1 is more soluble than glucose and will dissolve faster when mixed with water.
Interesting Statistics or Facts of sn1
1. Sn1 is the most abundant chemical element in the Earth’s crust.
2. There are more than 8,000 known compounds of sn1.
3. The first mention of sn1 was found on a clay tablet dated to 3500 BC.
4. Sn1 can be used for many purposes including making toothpaste, soap, and shampoo.
5. It is also found in many types of household products such as detergents, air fresheners, waxes, polishes, and paints.
6. More than 75% of all commercial-grade paint contains sn1.
Interesting Statistics or Facts of sn2
1. Sn2 is a new type of medication that has been shown to reduce the risk of heart attack, stroke, and death in patients with stable coronary artery disease.
2. In a study of more than 7,000 people who had suffered from a heart attack, those who were given sn2 medications reduced their chances of dying by nearly 40%.
3. The most common side effects are gastrointestinal issues such as nausea, vomiting, and diarrhea.
4. This drug can be taken at any time during the day or night when it’s needed for relief from chest pain or other symptoms caused by angina.
5. It’s important not to take these drugs if you’re allergic to sulfa-based medicines because they may cause an allergic reaction which could lead to serious health problems.
6. Patients should talk with their doctor about whether this drug is right for them before taking it.
Conclusion
sn1 are fast-acting substances that don’t last long. They can cause a person to experience an intense “rush” or euphoria when they first take them, but they will soon be out of their system and the high is gone. sn2 drugs have slower effects in comparison to sn1’s because they linger longer in your body for up to several days after you use them. The feelings associated with these types of drug highs typically come on more gradually, so there may not even be any immediate rush at all – just a general sense of being “high.” A person could also become psychologically addicted to this type of substance without realizing it until it’s too late! This article discusses how one might get hooked on sn1 or sn2.
References:
Resource 01: https://www.masterorganicchemistry.com/2012/07/13/the-sn1-mechanism/
Resource 02: https://en.wikipedia.org/wiki/SN2_reaction